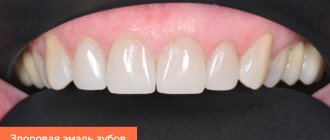
Composition of tooth enamel
Dental enamel is the most durable, wear-resistant and hard component in the human body. It represents the outer or surface layer of the teeth, completely covers its coronal and partially cervical part, and also performs protective functions. The main characteristics of enamel include:
- The color of tooth enamel varies from white to yellowish, and it can change shade (color) depending on a person’s dietary habits or bad habits.
- The thickest areas of enamel cover the chewing molars in the area of their anatomical tubercles; their thickness varies between 2.3-3.5 mm;
- The thinnest areas of tooth enamel are localized in paroxysmal areas (places of contact in their lateral projection), here the thickness of the protective layer reaches approximately 1.3 mm;
- The enamel that covers all the teeth in the human oral cavity is not capable of regeneration, because there are no living cellular structures in the tissues of this protective layer;
- Depending on the characteristics of each person’s body, up to 95% of the chemical composition of tooth enamel is presented in the form of mineral compounds. The remaining percentage is divided between water and organic matter in approximately a 2:1 ratio, respectively. In addition, depending on the percentage of mineral content of tooth enamel, it can be more or less transparent (the higher the percentage of minerals, the more transparent the enamel becomes).
Dentinoenamel junction
We will look at the dentinoenamel junction using the pictures below the text. In Fig. Figure 1 shows a tooth segment including dentin (D) and the inner enamel region (E), which is then shown in slightly enlarged form in Fig. 2.
In Fig. 2, a small section of the peripheral zone of dentin is highlighted to show its ultrastructural features directly below the dentin-enamel junction (DES) . On the exposed dentin surface, one can see branched terminal segments of Thoms fibers (TT), emerging from dentinal tubules (DT), surrounded by Neumann membranes (TN). This type of hypermineralized peritubular dentin is different from intertubular dentin (ID), which makes up the bulk of dentin. In a small area of specially decalcified dentin, i.e., without hydroxyapatite crystals, collagen microfibrils (CM) are shown embedded in the hard dentin ground substance.
In dentin, closer to the dentinoenamel junction , interglobular spaces (IS) are delimited by spherical structures of calcified dentin. They may contain some collagen microfibrils. There are no Neumann sheaths around the Thoms fibers that normally traverse the interglobular space. Some Toms fibers can penetrate short distances into the enamel (E). The dentin surface in the area of the dentinoenamel junction usually has a relief in the form of dentinal protrusions (DP), separated by ridges (D), located between groups of enamel prisms (EP). Each enamel prism in the group fits into the dentin so that they form small depressions (U) visible on the concave surface of each dentin projection. The lower left corner of this drawing shows an isolated enamel prism fitting into such a recess.
The contact zones of the enamel prisms with the dentin surface are visible in Fig. 3.
Other components in the enamel
In addition to the already mentioned main components of the enamel layer of the tooth, its chemical composition also contains a set of other components:
- Neonatal line - present exclusively on baby teeth, it looks like a dark-colored stripe (almost black). This line is located in the area of contact between two types of enamel, the first of which was formed before the baby was born, and the second after.
- Bundles and plates of dental enamel are special enamel formations containing prisms of a hypomineralized type, between which the interprismatic substance consists of the same material. It is noteworthy that the molecular structure of this material involves a large number of protein compounds. Many dentists are of the opinion that through the mentioned bundles and plates, various microorganisms penetrate into the enamel from the oral cavity, making their way to deeper dental tissues, causing caries, etc.
- Gunter-Schräger stripes are lines that stand out on the tooth enamel in a darker or lighter shade, the width of which does not exceed 100 microns. They are located perpendicular to the surface of the enamel layer and are formed as a result of opening its prisms.
- Retzius lines - in shape they resemble arches offset from the central one, located symmetrically in relation to each other. When cut across a tooth , these formations resemble rings inside a tree trunk. The formation of Retzius lines corresponds to different periods of mineralization of the enamel layer.
Harm of fluoride to teeth
The harm of fluoride, for teeth in particular and for the body as a whole, consists of two things - fluorosis and poisoning.
Fluorosis occurs when there is too much fluoride in the body. In the case of fluorosis, fluoride is dangerous even before teeth erupt; it disrupts the formation of their enamel. You can read more about fluorosis in the article “Dental Fluorosis.”
In adulthood, excess fluoride can lead to poisoning. Poisoning is likely due to an overdose of systemic (water, milk, salt, tablets) rather than local fluoride preparations. For young children who may not yet be able to control their swallowing, special children's pastes are produced. They have a much lower fluoride concentration (500 ppm), and very little of the paste itself is needed (“traces” or “the size of a pea”).
Features of the structure of the enamel of baby teeth
The main distinguishing feature of the enamel layer of children's teeth is that it is less durable and also much thinner than the enamel of permanent teeth. This is explained by the lower content of mineral compounds in teeth in relation to water and organic substances. Considering these features, if you examine baby teeth and their enamel layer under a microscope, you will notice the following differences:
- Due to the fact that the service life, as well as periods of mineralization and the tendency towards this process are shorter, the Retzius lines are much less pronounced in the structure of primary dental units.
- If in permanent teeth enamel prisms are located apically, then in milk teeth their direction is completely different, they are located horizontally.
- In children's primary teeth, the final enamel layer is much less pronounced; prisms are clearly visible on its surface, while its structure is much more porous, with microscopic cracks present.
Under the influence of each of these features, tooth enamel in children is more susceptible to wear and damage. For this reason, children develop caries much more often and it progresses faster, which is why it is important to visit the dentist regularly and have their teeth treated in a timely manner.
Fluoride preparations for teeth
There are many fluoride preparations for teeth. These are solutions, pastes, foams, gels, varnishes and even filling materials that emit fluoride. Different drugs are used in different conditions: some (pastes, gels, foams) are used at home, others (varnishes, filling materials) are used only in the dentist’s office.
The fluorine compounds in their composition are also different. Let's figure out which ones are better, more effective and worth looking for in toothpaste on the store shelf. For clarity, I will immediately focus on their pros and cons.
Fluorine compounds are inorganic and organic. The former are also divided into easily and slowly dissociating (releasing fluorine).
Inorganic easily dissociating fluorine compounds are sodium fluoride, tin fluoride, acid phosphate sodium fluoride.
Sodium fluoride is the most widely used.
| Pros + | Minuses - |
| quickly releases fluorine (like all compounds of this group) | Can only be combined with silicon abrasives (silica) |
| is well fixed in dental plaque and on the oral mucosa | quickly eliminated from the oral cavity |
| forms calcium fluoride (what is good about it, we said earlier) |
Tin fluoride
| + | – |
| tin has an antimicrobial effect | tastes bad |
| tin phosphate fluoride is formed and precipitated, which slows down the carious process | the same phosphate fluoride stains demineralized enamel and irritates inflamed gums |
| quickly breaks down in aqueous solutions |
Sodium acid phosphate fluoride (APF) is very common in the United States and Canada.
| + | – |
| in the acidic environment of APF, more calcium fluoride and HF acid are formed, which penetrates deep into the enamel | sour taste acid damages ceramic and composite restorations |
| phosphate enhances the penetration of fluoride into enamel |
Slowly dissociating inorganic fluorine compounds are neutral sodium monofluorophosphate and calcium fluoride.
Neutral sodium monofluorophosphate (Sodium monofluorophosphate) was widely used until 1985, because it was combined with the abrasives used at that time - chalk and pumice. Nowadays it can be rarely found due to many disadvantages:
| + | – |
| captured by oral bacteria | does not affect the formation of acid in dental plaque |
| does not form enough calcium fluoride | |
| quickly eliminated from the oral cavity | |
| short contact of fluoride with enamel due to its slow release and rapid removal of the compound itself |
Calcium fluoride is used in fluoride varnishes in high concentrations. It is non-toxic, easily incorporated into the micropores of the enamel, and in an acidic environment it releases fluoride and calcium ions.
Aminofluoride
The most popular organic fluoride compound is aminofluoride (Olaflur, Amifluor). It is the most effective at present, having many advantages:
| + | – |
| distributes well and stays on the enamel for a long time due to its surfactant structure | specific taste may stain teeth gray (with poor hygiene) high cost, complex production |
| forms calcium fluoride, which lasts much longer | |
| creates an acidic environment for better interaction of fluoride with enamel | |
| pronounced antimicrobial effect | |
| disrupts the adhesion of microorganisms to the tooth, slows down the formation and growth of dental plaque | |
| creates a fluoride depot in the deeper, subsurface layers of enamel |
Types of damage to tooth enamel
Over the course of life, even if you provide your teeth with proper care and follow the rules of oral hygiene prescribed by dentists, the enamel layer gradually wears out and is destroyed. This contributes to the occurrence of various diseases of the oral cavity; it is influenced by the food a person eats, etc.
Among the main causes of damage and destruction of tooth enamel, dentists identify:
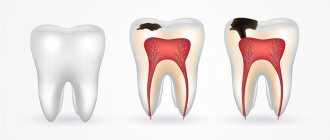
- Erosion is damage to the enamel layer, and then dentin, which is not associated with carious lesions of the teeth . The essence of this pathological process lies in disorders of mineral metabolism. As a result, disturbances occur in the crystalline structure of the enamel, which is manifested by its focal thinning and destruction. Externally, erosions look like local darkening on a round or oval tooth. The occurrence of erosions is provoked by the consumption of foods with high acidity levels, pathologies of the gastrointestinal tract, the use of certain medications, and the use of aggressive tooth powder or paste.
- Excessive sensitivity of tooth enamel - this disorder is especially pronounced in the form of painful sensations when the teeth cold or hot food, drinks, and even as a result of contact with cool air. The sensitivity of tooth enamel develops due to its thinning under the influence of the factors already described above. A thinned enamel layer puts teeth at increased risk of caries and other dental pathologies.
- Necrosis - this term characterizes multiple lesions of the hard tissues of the tooth, especially the enamel layer and dentin. The pathological process is initially expressed in the appearance of small light spots on the surface of the tooth , which subsequently darken and deepen. The progression of pathology threatens tooth destruction and is accompanied by a number of other oral diseases. The main reasons for the development of necrosis include gastrointestinal diseases, hormonal imbalances, metabolic disorders in the body, and work in hazardous industries.
- Caries is a carious lesion that threatens teeth , primarily affecting the enamel layer of the structure , gradually destroying it and spreading to deeper tissues. There are many reasons for the development of caries, from non-compliance with the rules of oral hygiene and irregular tooth brushing, to pathologies of the structures of the oral cavity, diseases of the gastrointestinal tract and metabolic disorders. If you start caries treatment at the stage when the lesions have affected only the enamel layer, you can only get by installing a filling or even restoring the enamel. But progressive caries is dangerous for teeth due to destruction, which may lead to the need for its complete removal.
- Mechanical damage - due to the fact that the main function of the enamel layer is to provide protection to the teeth , it primarily suffers from the effects of external adverse factors. Mechanical damage to the enamel includes cracks and other violations of its integrity due to blows, bruises, eating too hard food, etc. If the enamel of at least one tooth has been subjected to aggressive mechanical action, you should consult a doctor for an examination and, if necessary, subsequent treatment.
- Wedge-shaped defect – this term characterizes the pathological process in which the area of the dental neck is exposed. In such cases, the thinnest and most vulnerable areas of the enamel layer, located at the base of the teeth, are negatively affected. In addition to visible receding gums, damage to the enamel is indicated by a change in its color, as well as an acute reaction to hot and cold.
Organic and inorganic enamel matrices –
Tooth enamel is quite unique, and in many ways is absolutely unlike other hard tooth tissues. Firstly, enamel is the only dental tissue that comes from the ectoderm (the development of all others is associated with the mesoderm). Secondly, if the organic matrix of other dental tissues is formed mainly by collagen fibers, then the organic enamel matrix does not contain collagen and is formed by the so-called “enamel proteins”. Thirdly, hydroxyapatite crystals in tooth enamel are much larger than in other mineralized tooth tissues.
The last point is that other hard dental tissues continue to be synthesized by cells during the life of an individual (odontoblasts and cementoblasts, respectively), but in mature enamel there are no cellular elements, and therefore, after its maturation, no growth can occur. This is due to the resorption of enameloblast cells during the process of enamelogenesis.
1) Organic matrix of tooth enamel –
We have already said above that the organic matrix of tooth enamel consists of non-collagenous proteins (proteins), which are the product of secretion of enameloblasts, and they are called the term “enamel proteins”. The function of the organic matrix is to adsorb minerals, resulting in the formation of apatite crystals around the enamel proteins. However, as the enamel matures, the organic matrix is almost completely lost.
All enamel proteins are conventionally divided into four types - 1) enamelins and 2) amelogenins, 3) ameloblastins and 4) taftelins. Enamelins are acidic glycoproteins with a large molecular weight, which are characterized by a high content of glycine, serine, aspartic and gamma-carboxyglutamic acids. In turn, amelogenins are hydrophilic glycoproteins (2 times less molecular weight), enriched with proline, leucine, histidine and gamma-carboxyglutamic acid.
Ameloblastins and taftelins occur only during the period of amelogenesis (enamel formation). In addition to enamelins and amelogenins, the organic matrix of mature enamel also contains glycosaminoglycans, proteoglycans, as well as various classes of lipids. All these organic substances are one way or another involved in the processes of mineralization of the organic matrix (protein calcification).
2) Inorganic enamel matrix –
According to research by E.V. Borovsky, tooth enamel contains the following inorganic compounds (average values):
- hydroxyapatite [Ca10(PO4)6(OH)2] – 75.04%
- carbonate-apatite [Ca10(PO4)6(CaCO3)2] – 12.06%,
- chlorapatite [Ca10(PO4)6(Cl)2] – 4.39%,
- fluorapatite [Ca10(PO4)6(F)2] – 0.66%,
- calcium carbonate – 1.33%,
- magnesium carbonate – 1.62%.
These compounds contain about 37% calcium and about 17% phosphorus. Thus, the main mineral salt in the composition of enamel (as well as dentin and tooth root cement) is “calcium phosphate” in the form of apatite crystals, which will additionally contain either hydroxyl residues, or a carbonate group, or chlorine or fluorine. But in addition to these elements and compounds, lead, zinc, aluminum, copper, molybdenum, sodium, strontium, sulfur, tin and titanium are also included in enamel apatite crystals (in extremely small quantities).
In the superficial layers of enamel there are more apatite crystals containing fluorine, lead or zinc, but in the deep layers of enamel their content will be less. At the same time, on the contrary, there will be more apatite crystals containing sodium, magnesium or carbonates in the area of the enamel-dentin junction, and fewer in the surface layers of enamel (24stoma.ru). This "ion gradient" has a certain meaning. For example, apatites with inclusions of sodium, magnesium or carbonates are highly resistant to splitting along the enamel-dentin junction.
More superficially located apatites with inclusions of fluorine, lead and zinc - thanks to these elements, they acquire special strength and resistance to acids. Enamel containing such apatite crystals (such as fluorapatite) is significantly resistant to caries, because fluorapatite begins to degrade at a lower pH value compared to ordinary hydroxyapatite. For example, for ordinary hydroxyapatite the critical pH value will be 5.5, but for fluorapatite – pH 4.6.
In addition, it should be noted that enamel hydroxyapatite crystals (compared to dentin and cement) will have a significantly larger size. For example, in dentin, hydroxyapatite crystals are 20 nm long, 18-20 nm wide, and 3.5 nm thick, which indicates their small size and needle-shaped shape. In turn, in enamel, hydroxyapatite crystals look like hexagonal plates with a length of about 200 nm (but crystals with a size of 500 to 600 nm can also be found), a width of 40–90 nm and an average thickness of 25–40 nm.
Important: due to the absence of enameloblasts in mature enamel, enamel does not have the ability to regenerate (like tooth root cement or dentin). However, despite this, the inorganic enamel matrix is in the process of constant remodeling - thanks to the ongoing processes of mineralization / demineralization. Moreover, the entry of calcium, phosphorus, and fluorine ions into the enamel occurs not only from saliva, but also from the dentin and pulp of the tooth (thanks to the so-called “enamel spindles”).
Strengthening tooth enamel
Today in dental practice there are many effective ways to strengthen the enamel, which allows you to maintain its integrity and prevent destruction and diseases of the dentition. At the same time, methods of strengthening and protecting the enamel layer are divided into two groups, the first are intended for adults, the second for children.
Main functions
The main functional task of this part of the tooth is the reliable protection of its internal structures - dentin and pulp. It is a barrier that prevents irritating mechanical, thermal and chemical effects on more sensitive and susceptible tissues. It is thanks to its hardness that we are able to bite and chew food freely and painlessly.
Generally speaking, teeth also help us reproduce sounds and, accordingly, speak. They are also responsible for the attractiveness of our smile and appearance in general, which ensures our comfort both psychologically and socially.
Enamel helps you bite and chew food
Strengthening baby teeth enamel
As was said earlier in relation to baby teeth, their enamel is more vulnerable. To protect it, saving the child from premature loss of dental units and problems in the future, doctors perform the following actions to provide temporary protection:
- Fluoridation involves treating teeth with special fluoride-based compounds; it is recommended to repeat this procedure 2-3 times a year.
- Fissure sealing - the dentist performs the procedure of filling the recesses and grooves of chewing teeth with temporary filling material, protecting the dental structures from the negative effects of harmful microorganisms and other unfavorable factors.
- Application gels and preventive mouth guards for teeth - the method is based on enriching the enamel layer with useful components (fluorine, calcium, vitamins) through the use of special products.
Fluoride toothpaste
The most commonly used drug that contains fluoride in its composition is fluoride toothpaste. There are several tips to help maximize the positive effects of fluoride:
— toothpaste should be in the oral cavity for at least 2-3 minutes (time of brushing teeth);
— frequency of brushing teeth – 2 times a day;
- after brushing, you should rinse your teeth with water only once, “filtering” it between your teeth;
— at night, it is recommended to fill the interdental spaces with fluoride toothpaste, applying it to the dental floss.
Well, we talked about the benefits of fluoride for teeth. This is a scientifically substantiated, proven fact. But why then are toothpastes with the proud inscription “Fluoride Free” increasingly conquering the market? Perhaps fluoride is still harmful to teeth?
Strengthening the enamel of molars
There are more methods to preserve molars and maintain the condition of their enamel layer. Firstly, this is due to fewer contraindications for adults. Secondly, molars require long-term strengthening.
The main methods of strengthening the enamel of permanent teeth include:
- Drug therapy is based on the use of vitamin complexes containing vitamins of groups B6, B12, D. In addition, the patient is selected drugs that promote better absorption of calcium and fluoride by the body.
- Special gels and oral hygiene products – this technique uses specialized toothpastes and gels containing components necessary for teeth to strengthen and maintain the condition of the enamel layer. Also, teeth are subjected to unimaginative cleaning in a dental office.
- Mineralization and preventive cleaning – mineralization is performed using special means to increase the strength of the enamel and reduce its susceptibility to a number of negative factors. As for cleaning, such procedures are performed by dentists in the clinic using special equipment. During cleaning, plaque and tartar are eliminated , pathogenic bacteria and microorganisms that can harm the enamel layer are removed.
- Home prevention - to maintain healthy teeth and enamel, patients are advised to perform a light massage of the gums, enrich the diet with fresh vegetables and fruits rich in vitamins.
Author: Zhukov M.A.
How common is Priestley's plaque?
According to various sources, from 2.4 to 18% of people experience this plaque.
Plaque is found equally often in boys and girls.
The dependence of the frequency of plaque on the nature of nutrition and the level of oral hygiene is not yet clear.
Taking iron supplements by a child or even his mother during pregnancy makes plaque more likely.
Several authors have proposed their classifications of black plaque according to severity.
Encouraging fact: When the dentist removes plaque, there is usually good, strong, healthy enamel underneath. Most scientists believe that the presence of Priestley plaque even reduces the risk of tooth decay.
Studies have shown that the saliva of children with Priestley's plaque typically has a higher concentration of calcium and a buffer capacity - that is, the ability to resist acidification. Perhaps this fact explains the lower risk of caries.
As I mentioned, bacterial plaque doesn’t just affect children.
The photo below is a real clinical case, and not the actor’s preparation for filming the new part of Mad Max.
The photo shows a 60-year-old man with no bad habits and good oral hygiene.
The cause of his plaque turned out to be the bacterium Prevotella melaninogenica .
This bacterium may be part of the normal oral flora, giving the distinctive black color to plaque, especially in children. The bacterium produces hydrogen sulfide, which interacts with iron found in saliva. The result is black staining of the teeth and tongue.
In this case, she chose the oral cavity of an adult man.
By the way, this man still managed to get rid of plaque with the help of dentists and regular dental hygiene.
What can plaque on teeth be confused with?
Areas of enamel may appear yellowish due to severe erosion and dentin showing through
And teeth can also be green...