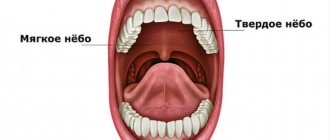
Dysgeusia (distortion of taste) is a disease in which the sense of taste is partially or completely absent. There are many reasons for this disease. Using the sense of taste, we can determine the taste of food and other elements entering the body. The sense organs that are responsible for this function are called taste buds.
They are located on the surface of the tongue. Under some conditions, a person may lose the ability to taste foods. Experts distinguish four main types of such disorders, namely pargeusia, dysgeusia, ageusia or hypogeusia. The complete absence of all taste sensations is called ageusia, partial weakening is called hypogeusia . Sometimes, a person constantly feels an unpleasant taste, regardless of the product he consumes. Distorted taste, which has a metallic taste, is a sign of dysgeusia.
Taste qualities with dysgeusia are distorted
Based on the above, taste qualities in dysgeusia are not completely absent, but are distorted. Some experts call dysgeusia (taste distortion) any taste disorder, while others use this term to mean a certain type of taste disorder. That is, dysgeusia is any change in taste, including a strange taste. No one is immune from the occurrence of taste deviations. They can occur at any age. People with dysgeusia constantly feel an unpleasant, salty, rancid or metallic taste in their mouth.
Even ice cream can have a salty or metallic taste. There are cases when people also experience changes in smell. Taste buds and receptors responsible for smell are interconnected, so this phenomenon is quite common.
Taste perception in patients with type 2 diabetes mellitus (literature review)
Diabetes mellitus remains one of the most dramatic problems in clinical medicine. First of all, this is due to its wide distribution, severity of complications, disability, high mortality, complexity of pathogenesis, and the lack of clear understanding of the causes and mechanisms of development of this disease to date.
Type 2 diabetes mellitus is called a multifactorial disease with an extremely complex etiology and pathogenesis, which is now being solved at the molecular gene level [29].
Statistical data from both domestic and foreign studies show that from 1% to 3% of the world's population suffers from this chronic lifelong pathology and their number is steadily increasing every year. Children have also become more likely to suffer from diabetes, and from a very early age [3, 6]. The annual increase in this disease is predicted to be approximately 5–7% [21, 23].
The manifestation of type 2 diabetes mellitus can be preceded by insulin resistance and hyperinsulinemia for decades. The problem of insulin resistance is particularly acute, as it is associated with the occurrence and development of severe micro- and macroangiopathies, damage to the function of the vascular endothelium [1, 5, 23].
Along with impaired secretion, insulin resistance is a major component of type 2 diabetes. Insulin resistance syndrome is manifested by hyperinsulinemia, dyslipoproteinemia, hypertension and decreased fibrinolysis, i.e., risk factors for non-insulin-dependent diabetes mellitus and cardiovascular diseases. The biguanide metformin has long been used to treat insulin resistance. Newer drugs include troglitazone, a thiazolidinedione drug. This drug significantly reduces insulin resistance, but sometimes has toxic effects, particularly on the liver [10].
Data obtained by Kekalainen et al. [23], indicate that hyperinsulinemia and a group of associated risk factors for cardiovascular pathology are important prognostic factors for the development of diabetes mellitus over 8 years, regardless of family history of diabetes mellitus with the regulation of local and systemic circulation.
Researchers provide evidence of an increase in myocardial infarction in patients receiving intensive insulin treatment, especially when their level of glycosylated hemoglobin decreases. It is indicated that oral hypoglycemic drugs that do not belong to the sulfonylurea group (troglitazone, metformin) may reduce the indications for transferring patients to insulin.
Today, the question of the advisability of intensive insulin therapy in patients with type 2 diabetes mellitus with macroangiopathy remains open and controversial [10, 14].
One of the main manifestations of diabetes mellitus is considered to be an increase in blood sugar levels, i.e. hyperglycemia. Most authors believe that it plays a major role in the development of diabetic complications.
It has been shown that in patients with diabetes mellitus, hyperglycemia causes an increase in the oxidative process, which leads to intensified lipid peroxidation of cell membranes [22]. These disorders initiate oxidative stress in the vessels, activating transcription factors in the cells of the vascular wall, which ultimately leads to the progression of atherosclerosis [8].
Gopaul et al. [20] concluded that oxidative stress is an early process in the progression of type 2 diabetes mellitus and may precede the development of endothelial dysfunction and insulin resistance.
According to James et al. [22] hyperinsulinemia is the main element uniting obesity, diabetes mellitus and arterial hypertension.
Huang et al. [21] identified a relationship between lipoprotein concentrations and blood coagulation parameters in elderly patients with type 2 diabetes mellitus, showing an increase in blood coagulation ability and a decrease in fibrinolysis, especially in those who had suffered a cerebral infarction in the past. These changes aggravate the course of diabetes mellitus and are directly related to the development of vascular complications of type 2 diabetes mellitus. The incidence of fatal outcomes reached 44.7% [19].
The data obtained suggest that in the blood of diabetic patients there is a fraction of highly atherogenic multimodified lipoprotein particles, which can play a significant role in the processes of accelerated atherogenesis.
Shah et al. [27] concluded that in hyperinsulinemia and hyperglycemia, increased levels of plasma free fatty acids cause disruption of glucose metabolism in women by inhibiting glucose utilization throughout the body, glucose uptake into muscles, and suppression of glucose production in internal organs. Thus, increased levels of free fatty acids cause disturbances in glucose metabolism in women.
Age is also an important factor in the development of diabetes. In old age, diabetes is in most cases quite mild and patients often do without insulin injections; in young people, diabetes is characterized by a more severe course, requiring daily insulin injections [3, 18, 22].
Uncontrolled type 2 diabetes mellitus is defined as persistent fasting hyperglycemia > 300 mg/dL or an increase in glycosylated hemoglobin levels above the upper limit of normal by 100% or more, repeated severe hypoglycemia, sharp fluctuations in glucose levels, repeated episodes of diabetic ketoacidosis, frequent absences from school, or work. Due to the decreased immune status in patients with diabetes, the percentage of infectious complications is quite high (ulceration of the lower extremities is 25%). Hospitalization is often necessary due to complications of diabetes mellitus from the cardiovascular system, nervous system, kidneys and other organs, as well as due to infection, surgery and other indications [6, 9, 24].
The age- and sex-adjusted relative risk of mortality from all causes was 1.51 for newly diagnosed diabetes according to American Diabetes Association criteria. A fasting glucose concentration of 7.0 mmol/L or higher regardless of plasma glucose concentration on an oral glucose tolerance test, 1.60 for newly diagnosed diabetes according to WHO criteria, and 1.98 for isolated hyperglycemia on an oral glucose tolerance test. glucose tolerance [32].
In summary, undiagnosed diabetes is associated with increased mortality from all causes using all criteria. Increased mortality due to cardiovascular disease is associated only with diabetes diagnosed by fasting glucose concentration. Mortality in this population was similar using American Diabetes Association and WHO criteria [32].
At the current level of development of diabetology, the use of high technologies and research methods at the molecular and genetic levels allows us to better understand the subtle molecular mechanisms of the pathogenesis of diabetes mellitus and especially its late complications [7].
Based on this, early diagnosis and prevention of diabetes, vascular and other complications should be carried out [2, 3].
In recent years, a significant increase in the life expectancy of patients with type 2 diabetes has been achieved, as well as a significant improvement in the quality of life of these people [6, 9, 15]. However, with the increase in the life expectancy of patients, a new, extremely important problem has come to the fore - the increase in the frequency and severity of late complications of the disease with damage to almost all organs and systems [6, 9].
The incidence of diabetes mellitus in the population also has a special social significance, which lies in the fact that it leads to early disability and mortality due to late vascular complications. These include: microangiopathy (retinopathy, nephropathy and gastropathy), macroangiopathy (myocardial infarction, stroke, gangrene of the lower extremities) [17].
Diabetic cardioneuronephro- and retinopathy still remain fatal complications of diabetes mellitus, as they inevitably lead to the death of patients from terminal renal and heart failure [24].
The most promising and economical direction for healthcare in the development of modern diabetes care is the prevention of vascular complications of diabetes mellitus, which is possible only with early diagnosis and timely prescription of pathogenetic therapy.
According to Stender S. et al. [28] 30–75% of diabetic patients have impaired gastrointestinal tract function. The incidence of gastrointestinal damage in diabetes can be largely explained by autonomic neuropathy, primarily damage to the vagus nerve. In this case, local disturbance of microcirculation, especially noticeable when teeth and gums are damaged, is of certain importance. Thus, clinical signs of diabetic periodontitis in the form of pasty and bleeding gums, multiple caries, dental plaque, loosening and loss of teeth, almost always accompanied by pyorrhea, are combined with microangiopathy in approximately 2/3 of patients with diabetes [3].
Also in these patients both the stomach and intestines are affected; diabetic enteropathy develops, leading to expansion of the stomach and disruption of the regulation of digestive processes, constipation and diarrhea; A serious complication associated with nervous disorders develops - sphincter atony and fecal incontinence. Diabetes mellitus does not initiate the occurrence of peptic ulcers. With poor compensation of diabetes mellitus, the normal functioning of the liver is disrupted and hepatomegaly develops [13, 14].
Diabetes is accompanied by diseases of the gastroduodenal system, which is determined by the severity of carbohydrate metabolism disorders. Disorders of metabolic processes in patients with diabetes mellitus are secondary in nature, which is associated with changes in the motor function of the stomach and duodenum, as well as with destructive processes in the mucous membrane. The autoimmune reaction to Helicobacter pylori in the mucous membrane of the stomach and duodenum underlies the dystrophic and necrobiotic processes leading to ulcerogenesis [13, 14].
The work carried out can serve as the basis for creating new approaches to the treatment of patients with type 2 diabetes mellitus, taking into account changes in the gastroduodenal system.
In the pathogenesis of diabetes, disorders of the nervous system, including sensory neuropathies, have been especially intensively studied in recent years [23]. As a rule, diabetes is characterized by multiple damage to a number of peripheral nerves (polyneuropathy), but in some cases diabetic mononeuropathy may develop [17, 19]. It develops as a result of damage to endoneurial vessels, which is confirmed by the presence of a relationship between the thickness of the basement membrane of these vessels and the density of nerve fibers in the peripheral nerve in diabetes mellitus. More than 50% of patients with diabetes suffer from diabetic polyneuropathy.
The frequency of damage to the nervous system in diabetes mellitus depends not only on the duration of the disease, but also on the age of the patients. Most often they appear in patients over 50 years of age after any long period of diabetes mellitus [3, 15].
In an attempt to uncover the reasons for the failure of therapy with antihyperglycemic drugs in adult patients with diabetes mellitus without obesity, Sa et al. [26] name two factors: autoantibodies to islet cells or reversible beta cell refractoriness. These studies showed that the failure of treatment with glucose-lowering drugs is due to the presence of autoantibodies and beta-cell desensitization. Beta cell autoantibodies may be present in up to 44% of patients. But in patients with autoantibodies, beta cells retain the ability to restore their function after a period of rest as a result of insulin therapy. The preservation of the response of beta cells with function restored as a result of insulin therapy to the action of glucose-lowering drugs is not known and requires study.
In the world literature, the role of specialized therapy for patients with type 2 diabetes mellitus is acutely raised, taking into account modern advances in clinical diabetology [18].
The most common is the use of metformin (Metfogamma), which reduces weight gain, reduces the need for insulin and improves glycemic control, improving quality of life. Treatment with metformin should be recommended when switching to insulin therapy for type 2 diabetes. It has been shown that metformin does not significantly affect blood pressure in patients with type 2 diabetes mellitus receiving intensive insulin therapy [14, 16]. This drug should be taken 1 tablet 1-2 times a day, during or after meals. Dosages of 500, 850 and 1000 mg are prescribed individually depending on the concentration of glucose in the blood.
The similarity of the glucose-lowering effects of pioglitazone, metformin and glimepiride in patients with newly diagnosed type 2 diabetes mellitus was revealed. The peculiarities of the action of glimepiride (Amaril) in relation to the rapid decrease in the concentration of glucose in the blood plasma, which is taken in the morning before meals, 1 tablet (initial dose 1 mg) under the control of blood glucose concentration, were noted. A beneficial effect of pioglitazone (a derivative of the thiazolidinedione series) on the concentration of fasting plasma glucose and fatty acids was also noted, which should be taken into account when choosing drugs individually [14]. Pioglitazone is prescribed orally once a day (regardless of meals). Monotherapy with pioglitazone 15–30 mg/day; if necessary, the dose can be increased to 45 mg/day.
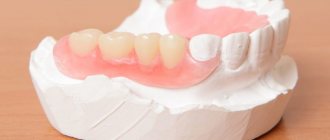
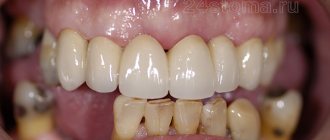
In diabetes mellitus, pathology of the oral cavity is often detected. Moreover, some lesions are associated with diabetes mellitus, others, although not so closely related to this disease, are nevertheless significantly more common among people with diabetes mellitus [11, 12].
The oral mucosa, in particular the tongue, indicates the state of health and primarily reflects the condition of the oral cavity and gastrointestinal tract [13]. The oral cavity is the earliest and most subtle indicator of various metabolic disorders in the body, and, therefore, studying the state of the oral mucosa in an age-related aspect can help in resolving some diagnostic and differential diagnostic problems [11, 12, 13].
The taste sensory system is defined today as a morphophysiological system that provides the perception and analysis of chemicals entering the oral cavity, and also reflects the functional state of the body.
A decrease in the number of taste buds leads to a decrease in taste sensitivity [11, 12]. In patients with type 2 diabetes mellitus, this is facilitated by impaired autoregulation of cerebral blood flow with autonomic neuropathy [11].
Loss of taste significantly reduces the quality of life and worsens the general and social state of human health [11, 12].
Italian researchers conducted a large-scale study on the etiology of taste disorders in the population. The authors found that taste disturbance is caused by a wide range of reasons: nervous, endocrine, metabolic, infectious, immunological, medicinal, surgical. The gustatory sensory system is vulnerable to both internal and external factors [25].
The activity of the taste sensory system is closely related to the functioning of the digestive apparatus. On the one hand, from the receptor section of the taste sensory system, the separation of saliva and gastric juice is reflexively “triggered”, influencing the motility of the digestive system; on the other hand, the level of activity of taste-perceiving structures depends on the degree of filling of the gastrointestinal tract with food. Thus, the sensitivity of taste buds, which is maximum on an empty stomach, decreases significantly after eating [25]. The gustatory sensory system plays a significant role in the preferential choice of certain food products [25]. When food loses taste and smell, as well as denervation of the oral cavity, destruction of the thalamic nuclei or the cortical part of the taste analyzer, taste analysis is partially or completely impaired [15].
In some cases, taste distortions are caused by diseases of internal organs and metabolic disorders; a feeling of bitterness is noted in diseases of the liver and gallbladder, a feeling of acidity in gastric dyspepsia, a feeling of sweetness in the mouth in severe forms of diabetes mellitus [4].
Against the background of the development of changes in the oral mucosa with symptoms of atrophy, increased keratinization of the epithelium and the appearance of erosions on the tongue, a progressive decrease in the number of functioning fungiform papillae and distortion of taste perception were noted [11, 12].
Many diseases of the mucous membrane of the oral cavity and tongue, being a reflection of any pathological processes in other organs, lead to changes in the functional state of the taste analyzer. At the same time, a change in taste sensitivity can be an early sign of a disease in the body [8]. But, despite the fact that these symptoms may have diagnostic significance, there are few works devoted to the study of the taste sensory system in this aspect [7].
Voros-Balog et al. [31] believe that “geographical” tongue (5.7%) may indicate an allergic disease. When examining the tongue of 1017 schoolchildren, in 35.11% of cases some changes in the language were found, including those due to diabetes [31].
In the activity of the taste analyzer, a significant role is played by such a food humoral factor as the content of glucose in the blood, which is the leading component of the nutritional biological need that determines the formation of the nutritional motivational state [7].
Disturbance of glucose homeostasis is a pathogenetic sign of diabetes mellitus. This disease affects people of different age and social groups, among which the number of young people increases every year. In the early stages of carbohydrate metabolism pathology, when a person feels healthy, the first signs of imbalance in clearly established glucose homeostasis and changes in the sensitivity of various perceptive structures, including the taste sensory system, may appear [7, 12].
To study taste perception in diabetes, along with the threshold method, the functional mobility method was used, and it was shown that impaired taste perception is correlated with the severity of the disease and blood sugar levels [7]. Peculiarities of taste sensitivity in diabetes mellitus in children have been noted [7].
Impaired perception processes in diabetes mellitus can also extend to the olfactory analyzer. Thus, Takayama et al. [30] describe acute loss of the sense of smell in a 61-year-old man with type 2 diabetes mellitus. After several years of successful treatment with diet (1600 kcal) and without medication, the symptoms of hyposmia disappeared. These authors believe that hyposmia was caused by diabetic mononeuropathy of the first cranial nerve (n. olfactorius).
The state of glucose homeostasis, maintained by the corresponding functional system, is one of the criteria for health and well-coordinated operation of the system. This position is confirmed by the study of taste perception in practically healthy people with intravenous administration of glucose, as well as by the study of the humoral background of the gastrolingual reflex [13].
Taking into account the data known in the literature on the role of the taste receptor apparatus in the activity of the whole organism, and especially in diabetes mellitus, the study of threshold density, determination of the functional mobility of the taste buds of the tongue can be used for diagnostic purposes for the prevention, early detection and timely treatment of neuropathy in patients diabetes mellitus type 2.
Literature
- Ametov AC Insulin secretion and insulin resistance: two sides of the same coin // Problems of endocrinology. 2002. Issue. 48, no. 3. pp. 31–37.
- Andreeva E.I. Daily blood pressure profile and optimization of arterial hypertension therapy in patients with diabetes mellitus: Author's abstract. dis. ...cand. honey. Sci. Rostov-on-Don. 2005. 24 p.
- Balabolkin M.I. Diabetology. M.: Medicine, 2000. 471 p.
- Balabolkin M.I. Insulin resistance and its significance in the pathogenesis of carbohydrate metabolism disorders and type 2 diabetes mellitus // Diabetes mellitus. 2002. No. 1. P. 12–20.
- Blagosklonnaya Ya. V., Babenko A. Yu., Krasilnikova E. I. Diabetes mellitus type II (non-insulin dependent) // New St. Petersburg Medical Gazette. 2001. No. 3. P. 17–23.
- Davydov A.L. Hemodynamic and metabolic disorders in patients with type 2 diabetes mellitus in combination with coronary heart disease: Abstract of thesis. dis…. Dr. med. Sci. M., 2000. 315 p.
- Zaklyakova L.V., Kurepova T.B. Vascular adhesion molecule-1 - a marker for early diagnosis of diabetic microangiopathies / Current issues in the diagnosis, treatment and prevention of the most common diseases of internal organs: Materials of the 4th therapeutic forum. Tyumen, 2005. pp. 16–17.
- Konyshev V. A. Food habits and vagaries of taste // Med. help. 2002. No. 1.S. 39–42.
- Mychka V. B., Gornostaev V. V., Chazova I. E. Cardiovascular complications of type 2 diabetes mellitus // Cardiology. 2002. No. 4. pp. 73–77.
- Naumenkova I.V. Clinical and diagnostic aspects of insulin resistance in patients with type 2 diabetes mellitus on insulin therapy: Author's abstract. dis. ...cand. honey. Sci. M., 2002. 20 p.
- Redinova T. L., Zlobina O. G., Merzlyakova O. G. Condition of the oral cavity in patients with diabetes mellitus / Modern issues in dentistry: Materials of the twelfth interregion. scientific-practical conf. dentists. Izhevsk State honey. acad. Izhevsk, 2000. pp. 148–152.
- Tokmakova S.I., Bondarenko O.V., Sysoeva O.V. The influence of age and metabolic disorders in diabetes mellitus on the oral mucosa / Dentistry. Ross. scientific forum with international with the participation of “Dentistry on the threshold of the third millennium”, February 6–9, 2001 MSMSU. M., 2001. pp. 117–118.
- Khvorostinka V.N., Krivonosova E.M. Pathogenetic features of the state of the gastroduodenal system in patients with diabetes mellitus // Medical practice. 2004. No. 3. P. 9–13.
- Belcher G., Schernthaner G. Changes in liver tests during 1-year treatment of patients with Type 2 diabetes with pioglitazone, metformin or gliclazide // Diabet. Med. 2005. Vol. 22, No. 8. P. 973–979.
- Ding R. et al. The effect of taste and consistency on swallow physiology in younger and older healthy individuals: a surface electromyographic study // J. Speech Lang Hear Res. 2003. Vol. 46, No. 4. P. 977–989.
- Douek IF, Allen SE, Ewings P., Cale EAM, Bingley PJ Continuing metformin when starting insulin I patients with Type 2 diabetes: A double-blind randomized placebo-control trial // Diabet. Med. 2005. Vol. 22, No. 5. P. 634–640.
- Duby II et al. Diabetic neuropathy: an intensive review // Health Syst Pharm. 2004. Vol. 61, No. 2. P. 160–173.
- Feinglos MN, Saasd MF, Pi-Sunyer EX Effects of liraglutide (NN2211), a long-acting GLP-1 anologue, on glucaemic control and body weight in subjects with Type 2 diabetes // Diabet. Med. 2005. Vol. 22, No. 8. P. 1016–1023.
- Gazis A., Pound N., Macfarlane R. Mortality in patients with diabetic neuropathic osteoarthropathy // Diabet. Med. 2004. Vol. 21, No. 11. P. 1243–1246.
- Gopaul NK, Manraj MD, Nebe A. Oxidative stress could precede endothelial dysfunction and insulin resistance in Indian Mauritians with impaired glucose metabolism // Diabetologia. 2001. Vol. 44, No. 6. P. 706–712.
- Huang S., Zhong R., Liang M. Relationship between lipoprotein (a) and activity of coagulation in old diabetic mellitus // China J. Mod. Med. 2004. Vol. 14, No. 17. P. 34–36.
- James AP, Watts GF, Mato JCL The effect of metformin and rosiglitazone on postprandial lipid metabolism in obese insulin-resistant subjects // Diabetes. Obesity and Metab. 2005. Vol. 7, 64. P. 381–389.
- Kekalainen P., Sarlund H., Pyorala K. Hyperinsulinemia cluster predicts the development of type 2 diabetes independently of family history of diabetes // Diabetes Care. 1999. Vol. 22, No. 1. P. 86–92.
- Rachmani R., Slavachevski I., Berla M. Teaching and motivating patients to control their risk factors retards progression of cardiovascular as well as microvascular seguelae of Type 2 diabetes mellitus—a randomized prospective 8-year follow-up study // Diabet. Med. 2005. Vol. 22, No. 4. P. 410–414.
- Rossig H. et al. Haloperidol and clozapine antagonize amphetamine disruption of latent inhibition of conditioned taste aversion //Psychopharmacology. 2003. Bd. 107, No. 3. S. 263–270.
- Sa JR, Silva R. C, Nasri E. Non-obese adult onset diabetes with oral hypoglycemic agent failure: Islet cell autoantibodies or reversible beta cell refractoriness // Braz. J. Med. and Biol. Res. 2003. Vol. 36, No. 10. P. 1301–1309.
- Shah PV Elevated free fatty acids impair glucose metabolism in women // Diabetes. 2003. Vol. 52, No. 1. P. 38–42.
- Stender S., Schuster H., Barter R. Comparison of rosuvastatin with atorvastatin, simvastatin and pravastatin in achieving cholesterol goats and improving plasma lipids in hypercholesterolaemic patients with or without the metabolic syndrome in the MERCURY I trail // Diabetes, Obesity and Metab, . 2005. V. 7, No. 4. P. 430–438.
- Tabaei BP, Engelgau MM, Herman WH A multivariate logistic regression equation to screen for dysglycaemia: Development and validation // Diabet. Med. 2005. Vol. 22, No. 5. P. 599–605.
- Takayama S., Sasaki T. Acute hyperemia in type 2 diabetes // J. Int. Med. Res. 2003. Vol. 31, No. 5. P. 466–468.
- Voros-Balog T., Vincze N., Banoczy J. Prevalence of tongue lesions in Hungarian children // Oral-Dis. 2003. Vol. 9, No. 2. P. 84–87.
- Wild SH, Smith EB, Lee AJ Criteria for previously undiagnosed diabetes and risk of mortality: 15-year follow-up of the Edinburgh Artery Study cohort // Diabet. Med. 2005. Vol. 22, No. 4. P. 490–496.
D. A. Kahramanova A. L. Davydov , Doctor of Medical Sciences, Professor V. A. Olkhin , Doctor of Medical Sciences, Professor MGMSU, Moscow
Contact information for authors for correspondence
Prevention of sweet moonshine
From the theory it becomes clear that it is necessary to minimize the appearance of glycerin and diethyl alcohol in the mash during the fermentation process and try to cut off these substances during distillation. To do this you need:
- Use mash water of normal acidity, preferably bottled.
- Reduce the fermentation period to a minimum by making the correct hydromodulus (1:4 or 1:5) and maintaining an optimal temperature of 26-29 °C.
- Avoid contamination of the wort with lactic acid bacteria.
- Change the yeast (if the problem of sweet moonshine has occurred before).
- Try to do the first distillation quickly so that as few harmful substances as possible appear during the boiling process.
- Perform distillation and rectification correctly, adhering to optimal temperature conditions, as well as selecting a sufficient number of “heads” and “tails”.
- Monitor the cleanliness of equipment (especially copper), do regular washing and cleaning.
- Use mash and distillation columns capable of normal purification of alcohol (correct design, sufficient drawer length, tightly packed SPN or RPN nozzle, cooler of suitable power, heater with smooth power control, automatic temperature control, etc.).