Pharmacological properties of the drug Cifran st
Pharmacodynamics. The pharmacological properties of the drug are determined by the properties of its active components. Ciprofloxacin, like other fluorinated quinolones, inhibits the enzyme DNA gyrase of bacteria, as a result of which bacterial DNA replication is disrupted. Ciprofloxacin is active against a wide range of microorganisms, gram-positive and gram-negative bacteria. The spectrum of action of ciprofloxacin covers the following microorganisms: aerobic gram-negative bacteria Escherichia coli, Klebsiella pneumoniae, Salmonella typhi, Proteus mirabilis, Proteus vulgaris, Shigella sonnei, Shigella boydii, Shigella dysenteriae, Shigella flexneri, Enterobacter cloacae, Moraxella morganii, Providencia rettgeri, Providencia stuartii , Citrobacter diversus, Citrobacter freundii, Serratia marcescens, Campylobacter jejuni, Pseudomonas aeruginosa, Neisseria gonorrhoeae, Haemophilus influenzae, Haemophilus parainfluenzae, Bacillus anthracis., Moraxella catarrhalis ; aerobic gram-positive bacteria - Enterococcus faecalis (some strains are moderately sensitive), Staphylococcus aureus (only methicillin-sensitive strains), Staphylococcus epidermidis (only methicillin-sensitive strains), Staphylococcus saprophyticus, Streptococcus pneumoniae (only methicillin-sensitive strains), Streptococcus pyogenes . Ciprofloxacin demonstrates in vitro minimal inhibitory concentration (MIC) of ≤1 µg/ml against the majority (90%) of the following specified microorganisms; however, the safety and effectiveness of ciprofloxacin in the treatment of infections caused by these microorganisms has not yet been established in adequate and well-controlled clinical trials: aerobic gram-positive bacteria: Staphylococcus haemolyticus, Staphylococcus hominis, Streptococcus pneumoniae (penicillin-resistant strains only); aerobic gram-negative microorganisms: Acinetobacter lwoffi, Aeromonas hydrophila, Edwardsiella tarda, Enterobacter aerogenes, Klebsiella oxytoca, Legionella pneumophila, Pasteurella multocida, Salmonella enteritidis, Vibrio Cholerae, Vibrio parahaemolyticus, Vibrio vulnificus, Yersinia enterocolitica . Most strains of Burkholderia cepacia and some strains of Stenotrophomonas maltophilia are resistant to ciprofloxacin, as are most anaerobic bacteria, including Bacteroides fragilis and Clostridium difficile . Tinidazole is a synthetic drug of the nitroimidazole group that has an antibacterial (relative to obligate anaerobic bacteria) and antiprotozoal effect. The mechanism of action of tinidazole is due to a violation of the DNA synthesis of microorganisms. Tinidazole is bactericidal. The spectrum of action of tinidazole covers the following microorganisms: anaerobic bacteria Bacteroides fragilis, Bacteroides melaninogenicus, Bacteroides spp., Clostridium spp., Eubacterium spp., Fusobacterium spp., Peptococcus spp., Peptostreptococcus spp., Vellionella spp., Gardnerella vaginalis ; Helicobacter pylori. Tinidazole is also active against protozoa: Trichomonas vaginalis, Entamoeba histolytica and Giardia lamblia . Pharmacokinetics. Not studied.
Indications for use of the drug Tsifran st
Therapy for mixed infections caused by aerobic and anaerobic microorganisms sensitive to the drug, as well as for the treatment of protozoal infections:
- respiratory tract infections: pleurisy, pleural empyema, lung abscess;
- ENT infections: chronic sinusitis, mastoiditis;
- skin and soft tissue infections: infected ulcers, abscesses, cellulitis, soft tissue infections in patients with diabetes;
- treatment of diarrhea, dysentery, mixed amoebic-bacterial infection of the gastrointestinal tract;
- intra-abdominal infections, including postoperative ones, caused by aerobic-anaerobic microbial associations;
- gynecological infections caused by aerobic-anaerobic microbial associations;
- bone infections: chronic osteomyelitis;
- orodental infections.
Tsifran ST
Tsifran ST (tinidazole + ciprofloxacin) is a combined antibacterial drug. Used for the treatment of combined aerobic and anaerobic invasions, as well as bacterial invasions of the gastrointestinal tract (chronic inflammation of the sinuses, abscess pneumonia, pyothorax, infections that develop when the anatomical barriers of the gastrointestinal tract are violated, gynecological infections, infections after surgical interventions, purulent-necrotic process in the bone tissue, dermatological infections, oral infections, amoebic and/or bacterial diarrhea and dysentery). Anaerobes - clostridia, bacteroides, peptococci and peptostreptococci - are sensitive to tinidazole. In most cases, in the presence of an anaerobic infection, aerobes are also present in the lesion, therefore, to increase efficiency, an antibacterial drug effective against aerobes is added. In Cifran ST, this is ciprofloxacin, sensitivity to which is shown by such aerobes as Escherichia coli, Klebsiella, Salmonella, Yersinia, Shigella, Haemophilus influenzae, Neisseria, mycoplasma, Vibrio cholerae, staphylococcus, streptococcus, chlamydia, mycoplasma, legionella, mycobacterium tuberculosis. Tinidazole and ciprofloxacin are rapidly absorbed from the digestive tract. Maximum concentrations of both substances are observed 1-2 hours after administration. The half-life of tinidazole is 12-14 hours. The drug has good penetrating ability.
It is excreted mainly in urine and in small quantities in feces. The presence of food contents in the gastrointestinal tract slows down the absorption of ciprofloxacin. Half-life is 3.5-4.5 hours. Cifran ST is not used in persons with individual intolerance to tinidazole and/or ciprofloxacin, as well as any other fluoroquinolone or imidazole antibiotics, or organic lesions of the nervous system. In pediatrics, Tsifran ST is not used. It is not recommended to use the drug in pregnant women and nursing mothers. The optimal time to take the drug is after meals. The structure of the tablet does not imply its breaking, chewing or any other violation of integrity. The tablet should be taken with a sufficient amount of water so that it can easily reach the site of absorption. The frequency of taking the drug is twice a day. The number of tablets per dose is determined by the dose of the drug: 2 tablets (for a dose of 250/300 mg) and 1 tablet (for a dose of 500/600 mg). During the drug course, it is recommended to avoid prolonged exposure to the sun in order to avoid phototoxicity reactions (if any develop, it is necessary to immediately interrupt pharmacotherapy). It is strictly not recommended to combine Tsifran ST with alcohol consumption, because ethanol in combination with tinidazole can provoke gastrointestinal spasms and dyspeptic reactions in the form of nausea and vomiting. During the course of medication, it is recommended to periodically do blood tests.
Use of the drug Cifran st
The usual dose of Tsifran ST for adults and children over 16 years of age is 1 tablet 2 times a day. Patients with creatinine clearance ≤20 ml/min, elderly patients and patients with low body weight are prescribed half the usual dose. The course of treatment depends on the severity of the infection, clinical dynamics and bacteriological examination data. Typically, the duration of therapy for acute infections is 5-7 days, in the case of treatment of chronic recurrent infections - 10-14 days. It is necessary to continue using the drug for 2 days after the symptoms of the disease disappear.
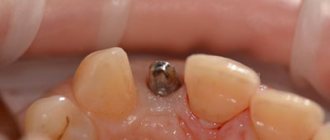
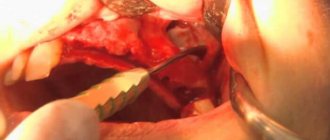
Necessary manipulations after tooth extraction:
- We apply something cold for the first 2 hours after surgery, from the outside to the removal site, after wrapping it in a paper napkin. If there is no special medical “cold”, then you can use any frozen product from the freezer.
- Exclude hot food for the first day, chew on the opposite side. Eliminate baths, saunas, hot baths and workouts for 5 days!
- Take medications prescribed by your doctor, usually these are 3 oral medications :
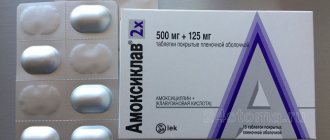
- Antibiotic - Amoxiclav 625 mg (1 tablet 2 times a day), or Tsifran-ST 0.5 mg (1 tablet 2 times a day).
This type of medicine is necessary after tooth extraction in order to protect the mucous membrane damaged after extraction from microbial infection.
- Antihistamine - Suprastin, Zodak, Tavegil (optional) - take according to instructions. These types of medications often have side effects such as drowsiness! Please take this into account when accepting.
This type of medication is necessary so that postoperative swelling goes away much faster.
- NSAIDs (non-steroidal anti-inflammatory drugs) for pain - Ketanov, Ketonal-Duo, Nimesil, Nise, etc. Take as directed. It is not recommended to take aspirin! If you have ever had stomach problems (gastritis, gastric ulcer or duodenal ulcer), it is recommended to take nimesil from this list of drugs! If you currently have such diseases, it is recommended to additionally drink omeprazole 20 mg 1 tablet in the morning while you are taking NSAIDs - this will protect your stomach lining from the side effects of NSAIDs.
This type of medication is necessary after tooth extraction to relieve pain and reduce postoperative inflammation.
4. Make oral baths after meals for 5-7 days with a solution of Chlorhexidine 0.05%. Those. take this solution into your mouth, tilt your head to the side where the operation was performed so that the solution flows into the cavity of the wound, hold it for 30 seconds and spit it out.
5. Application of Metrogil Denta ointment to the postoperative hole, suture line 2-3 times a day, for 5 days. There are several application options: With a cotton swab, a clean finger, or on a gauze ball.
Check your recommendations with the data and take what your doctor has prescribed for you.
And if you have any questions after your tooth extraction, always call us right away. We work around the clock - so we will always advise and advise you.
After 4 months, you can call us and make an appointment for a free consultation with an implant surgeon in order to regain a lost tooth using implant prosthetics, read more about this here.
Side effects of the drug Cifran st
Symptoms are usually mild or moderate and transient; only in isolated cases does treatment need to be discontinued. The most commonly reported symptoms were nausea, diarrhea, changes in laboratory parameters (ALT, AST), vomiting and skin rashes. Side effects that were observed rarely or in isolated cases (if it was impossible to exclude a connection between taking the drug and the resulting reactions): from the gastrointestinal tract : metallic taste or other taste disturbances, nausea, vomiting, soreness of the oral mucosa, dry mouth, thirst, hypersalivation , oral candidiasis, discoloration of the tongue, coated tongue, dysphagia, stomatitis, cholestatic jaundice, hepatitis, flatulence, constipation, diarrhea, anorexia, intestinal perforation, dyspeptic symptoms, salivation, pancreatitis, liver failure, liver necrosis, abdominal pain/discomfort, pseudomembranous colitis, gastrointestinal bleeding; from the central nervous system : headache, dizziness, nystagmus, insomnia, emotional disturbances, anxiety, convulsions (epistatus, etc.), increased intracranial pressure, hallucinations, confusion, nightmares, psychotic reactions (manic, phobias, depersonalization), agitation, delirium, irritability, depression, paresthesia, peripheral neuropathy, epileptic seizures, tremor, ataxia, coma, depression, drowsiness, lethargy, migraine, tic; from the cardiovascular system : palpitations, rhythm disturbances (including extrasystole, atrial flutter, ventricular ectopia, pirouette syndrome, tachycardia), increased or decreased blood pressure, syncope, angina pectoris, myocardial infarction, cardiogenic shock, thromboembolic complications, vasculitis; from the hematopoietic system : eosinophilia, leukopenia, neutropenia, thrombocytopenia, agranulocytosis, thrombocytosis, anemia (including hemolytic), bleeding, monocytosis, leukocytosis, severe depression of bone marrow function, methemoglobinemia; from the musculoskeletal system : pain in the limbs, arthralgia, myalgia, myasthenia gravis, myoclonus, arthritis, pain or dysfunction of the joints, tendon damage, tendinitis, gout attack, pain in the back, neck or chest; from the urinary system : interstitial nephritis, nephritis, secondary renal failure, polyuria, urinary retention, bleeding from the urethra, vaginal candidiasis and candiduria, vaginitis, discomfort in the mammary glands, crystalluria, cylindruria, hematuria, albuminuria, moniliasis, exacerbation of urolithiasis, darkening of urine; from the respiratory tract : dyspnea, nosebleeds, laryngeal or pulmonary edema, pharyngitis, bronchospasm, hiccups, hemoptysis; from the immune system and skin : allergic skin reactions, itching, vasculitis, rash, redness of the skin, urticaria, photosensitivity, fever, catarrhal phenomena, angioedema, conjunctivitis, skin candidiasis, hyperpigmentation, erythema nodosum, increased sweating, anaphylactic reaction (including including anaphylactic shock), toxic epidermal necrolysis, erythema multiforme, exfoliative dermatitis, Stevens-Johnson syndrome; from laboratory parameters : increased levels of alkaline phosphatase, amylase, lipase, LDH, liver transaminases (ALAT and AST), bilirubin, transient increase in creatinine and potassium in the blood plasma, increase in cholesterol and/or TG in the blood serum, prolongation or decrease in prothrombin time; from the senses : blurred vision, blurred vision, decreased visual acuity, diplopia, eye pain, chromatopsia, tinnitus, hearing loss, impaired sense of smell; other : general weakness, lymphadenopathy, petechiae, gynecomastia, dark urine, hyperglycemia, hyperesthesia, hypoesthesia, hypoglycemia.
Special instructions for the use of the drug Cifran st
During treatment with the drug you should not drink alcohol, as there is a risk of developing a disulfiram-like reaction. Cifran ST is prescribed with caution to patients with liver failure and acute porphyria. During treatment, sun exposure should be avoided. Patients taking Cifran ST must consume sufficient fluids to avoid crystalluria. Due to the possibility of a stimulating effect of ciprofloxacin on the central nervous system, Cifran ST should be prescribed with caution to patients with central nervous system disorders (cerebral atherosclerosis, epilepsy). Treatment should be interrupted in the presence of serious central nervous system disorders such as depression or psychosis. If the patient has anamnestic data regarding the noted defects in glucose-6-phosphate dehydrogenase activity in the family, one of the manifestations of which may be a hemolytic reaction to quinolines, the drug should be used with caution. There have been reports of tendon damage in patients taking quinolines; the risk of this damage is increased in elderly people and in case of simultaneous use of corticosteroids. The drug should be discontinued if the patient complains of pain or signs of inflammation or damage to the tendon, and rest is recommended until the diagnosis of tendonitis or tendon rupture is ruled out. In isolated cases, symptoms of sensory or sensorimotor polyneuropathy were noted in patients taking fluoroquinolones; in this case or if other neurological symptoms occur, the drug should be discontinued. The development of phototoxicity also requires discontinuation of therapy. When using broad-spectrum antibiotics, there is a risk of developing pseudomembranous colitis, the likelihood of which is higher in patients with severe persistent diarrhea. These cases have been reported very rarely with ciprofloxacin, but if pseudomembranous colitis is suspected, the drug should be discontinued and appropriate treatment (eg, oral vancomycin) should be initiated. If hypersensitivity reactions occur during the use of the drug, Cifran ST is discontinued. Treatment with Cifran ST must be carried out with constant monitoring of hematological parameters, since, as with any drug of the nitroimidazoles group, leukopenia and neutropenia may occur. When prescribing the drug to patients with severe liver pathology, dosage adjustment may be required. The drug should be discontinued at the first appearance of skin rashes or any other sign of hypersensitivity. Seizures and peripheral neuropathy (numbness or paresthesia of the extremities) have been reported in patients taking nitroimidazole drugs, including tinidazole and metronidazole. The appearance of neurological pathology requires urgent cessation of treatment with tinidazole. The drug should be prescribed with caution to patients with central nervous system diseases. Persons with reduced renal function require adjustment of the dosage regimen. During simultaneous treatment with Cifran ST and barbiturates, it is necessary to monitor heart rate, blood pressure and ECG. Impact on the ability to drive vehicles and operate dangerous machinery. When using the drug, you should refrain from driving vehicles and working with potentially dangerous mechanisms.
Types of antibiotics used in dentistry
To eliminate the symptoms and consequences of dental diseases, antibiotics of the following pharmacological groups can be used:
- Penicillins. The largest category of antibacterial agents. They are characterized by a wide spectrum of action and effectively destroy pathogenic microorganisms. They can be injected or used in tablet form.
- Macrolides. Remedies that are often used for diseases of the ENT organs. Dentists prescribe them only if the patient has poor tolerance to other antibacterial drugs or the pathogen is insensitive to penicillins.
- Cephalosporins. Medicines exhibiting excellent bactericidal properties. In dental practice, two representatives of the group are most often used - Cefaclor and Cephalexin.
- Lincosamides. Medicines that effectively control the inflammatory process. With their help, you can quickly eliminate inflammation and prevent further progression of dental complications. A popular representative of lincosamides is Lincomycin. It can be bought in the form of tablets, injections, ointments. The latter is suitable for tamponade of the inflamed socket.
- Fluoroquinols. Contains tsifran - a component that quickly destroys pathogenic flora. Indicated for purulent complications.
- Aminoglycosides. They are prescribed after extensive dental operations, if the wound is infected or the infection has entered the blood.
- Tetracyclines. They act by blocking protein synthesis in microbial cells. These include Oletetrin, Doxycycline. It should be noted that today tetracyclines are used less and less, since many modern infections are little sensitive to their active substance.
Interactions of the drug Cifran st
Serious and sometimes fatal reactions have been reported in patients taking ciprofloxacin and theophylline concomitantly. Since ciprofloxacin is a cytochrome P450 inhibitor, its combined use with other drugs metabolized mainly through this enzyme (for example, theophylline, methylxanthines, tizanidine) leads to an increase in the concentration of these drugs in the blood plasma and can cause corresponding toxic effects. If it is necessary to use theophylline and ciprofloxacin simultaneously, the level of theophylline in the blood serum should be monitored and the dose of the drug should be adjusted. Cifran ST should be taken no earlier than 4 hours after taking medications containing magnesium, aluminum, iron, sucralfate, and zinc. Ciprofloxacin affects the metabolism of caffeine, increasing its half-life. The following drug interactions are possible with the combined use of Tsifran ST and the following drugs:
A drug | Effect |
| Phenytoin | Change (increase or decrease) in serum levels, prolongation of half-life |
| Sulfonylureas | Hypoglycemia |
| Warfarin | Strengthening the anticoagulant effect |
| Methotrexate | Increased toxicity |
| NSAIDs | Changes in the central nervous system |
| Lithium preparations | Increased serum lithium levels |
| Cyclosporine, tacrolimus | Increased serum drug levels |
| Fluorouracil | Decreased clearance with increased toxicity |
| Liver enzyme inducers (eg phenobarbital, rifampicin) | Acceleration of elimination of tinidazole from blood plasma |
| Liver enzyme inhibitors (eg cimetidine, ketoconazole) | Slowing down the elimination of tinidazole from blood plasma |
| Sulfonylureas | Hypoglycemic effect |
| Cholestyramine | Reduced bioavailability of tinidazole |
| Probenecid | Decreased serum ciprofloxacin levels |
It is necessary to avoid drinking alcoholic beverages and medications containing ethyl alcohol or propylene glycol during treatment with Cifran ST and for another 3 days after that due to the possibility of intestinal spasms, nausea, vomiting, headaches and hot flashes. The development of psychotic reactions has been reported in alcoholics concomitantly taking metronidazole and disulfiram. Although similar reactions have not been reported with tinidazole, tinidazole should not be administered to patients taking disulfiram for 2 weeks the previous day.
CIFRAN OD
Interaction
Due to inhibition of microsomal enzymes in the liver, it increases the concentration of i.
prolongs T1/2 of theophylline and other xanthines (for example, caffeine), oral hypoglycemic drugs (for example, glibenclamide), indirect anticoagulants (for example, warfarin and its derivatives). If it is necessary to use it together with drugs of these groups, it is necessary to monitor the concentration of the drug in the blood and adjust the dosage regimen accordingly. In the presence of antacids containing magnesium hydroxide or aluminum hydroxide, the absorption of ciprofloxacin is reduced. Thus, the simultaneous use of these drugs should be excluded. In such cases, ciprofloxacin should be taken either 1-2 hours before or 4 hours after taking these drugs. Didanosine reduces the absorption of ciprofloxacin. This is due to the formation of complexes with magnesium salts contained in didanosine preparations.
When used together with probenecid and other drugs that block tubular secretion, the renal excretion of ciprofloxacin decreases.
Metoclopramide accelerates the absorption of the drug, which leads to a decrease in the time it takes to reach Cmax.
Co-administration of uricosuric drugs leads to a slower elimination (up to 50%) and an increase in plasma concentrations of ciprofloxacin.
When combined with other antimicrobial drugs (beta-lactam antibiotics, aminoglycosides, clindamycin, metronidazole), synergism is usually observed; can be successfully used in combination with azlocillin and ceftazidime for infections caused by Pseudomonas spp.; with mezlocillin, azlocillin and other beta-lactam antibiotics - for streptococcal infections; with isoxazolylpenicillins and vancomycin - for staphylococcal infections; with metronidazole and clindamycin - for anaerobic infections. Ciprofloxacin enhances the nephrotoxic effect of cyclosporine. There is an increase in serum creatinine concentration. In such patients, it is necessary to monitor this indicator 2 times a week.
Concomitant use of tizanidine with ciprofloxacin, which is an inhibitor of the CYP1A2 isoenzyme, leads to a 10-fold increase in the AUC of tizanidine. The result of combined use may be a clinically significant and prolonged decrease in blood pressure, leading to drowsiness, dizziness, and inhibited psychomotor reactions.
In patients receiving therapy with ciprofloxacin and phenytoin, variability (decrease or increase) in the concentration of phenytoin in the blood plasma was noted.
Combined use with nonsteroidal anti-inflammatory drugs (NSAIDs) increases the likelihood of side effects of ciprofloxacin on the central nervous system (risk of seizures).
Absorption of ciprofloxacin when administered orally decreases after cytotoxic therapy with antitumor and immunosuppressive drugs.
Overdose of the drug Cifran st, symptoms and treatment
Symptoms: Nephrotoxicity has been reported in cases of acute overdose. Treatment: empty the stomach by inducing vomiting, or rinse the stomach. The patient should be closely monitored and receive supportive care, including monitoring of renal function and administration of antacids containing magnesium, aluminum or calcium, which may reduce the absorption of ciprofloxacin. Adequate water regime should be maintained. Only a small amount of ciprofloxacin (≤10%) is removed from the body by hemodialysis or peritoneal dialysis.