Home | About us | Delivery | Advertisers | Login | Registration
- Medicines
- dietary supplementsVitamins
- Categories from A to Z
- Brands from A to Z
- Products from A to Z
- Medical equipment
- beauty
- Child
- Care
- Honey products appointments
- Herbs and herbal teas
- Medical nutrition
- Journey
- Making medicinesStock
Pharmacy online is the best pharmacy in Almaty, delivering medicines to Almaty. An online pharmacy or online pharmacy provides the following types of services: delivery of medicines, medicines to your home. Online pharmacy Almaty or online pharmacy Almaty delivers medicines to your home, as well as home delivery of medicines in Almaty.
my basket
Apteka84.kz is an online pharmacy that offers its customers medicines, medicinal and decorative cosmetics, dietary supplements, vitamins, baby food, intimate products for adults, medical equipment and thousands of other medical and cosmetic products at low prices. All data presented on the Apteka84.kz website is for informational purposes only and is not a substitute for professional medical care. Apteka84.kz strongly recommends that you carefully read the instructions for use contained in each package of medicines and other products. If you currently have any symptoms of the disease, you should seek help from a doctor. You should always tell your doctor or pharmacist about all the medicines you take. If you feel you need further help, please consult your local pharmacist or contact our GP online or by telephone.
© 2021 Pharmacy 84.
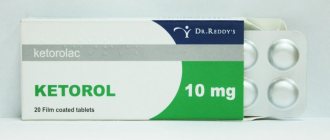
Ketorol Insta tablets dispersible in the oral cavity 10 mg No. 10x2
Name
Ketorol insta
Description
Light yellow, round, flat tablets engraved with an “I” on one side and smooth on the other.
Main active ingredient
Ketorolac tromethamine - 10 mg.
Release form
pills
Dosage
10 tablets in a blister made of aluminum/desiccant/aluminum foil, 1 blister in a cardboard box with instructions for use.
special instructions
pharmachologic effect
Non-steroidal anti-inflammatory drugs. ATX code: M01 AB15.
Pharmacodynamics
Ketorolac, being a non-steroidal anti-inflammatory drug, has an analgesic, antipyretic and anti-inflammatory effect. The mechanism of action at the biochemical level is inhibition of the cyclooxygenase enzyme, mainly in peripheral tissues, resulting in inhibition of the biosynthesis of prostaglandins - modulators of pain sensitivity, thermoregulation and inflammation. Ketorolac is a racemic mixture
—
S and
+
P enantiomers, with the analgesic effect due to
—
S shape. The drug does not affect opioid receptors, does not depress respiration, does not inhibit intestinal motility, does not have a sedative or anxiolytic effect, does not cause drug dependence, and does not affect the progression of the disease. Ketorolac inhibits platelet aggregation and increases bleeding time. The functional state of platelets is restored 24-48 hours after discontinuation of the drug.
Pharmacokinetics
Ketorolac tromethamine is rapidly and completely absorbed after oral administration with a peak plasma concentration of 0.87 mcg/ml 50 minutes after a single 10 mg dose. In healthy volunteers, the terminal plasma half-life averages 5.4 hours. In older people (average age 72 years) it is 6.2 hours. More than 99% of ketorolac in blood plasma is protein bound. In humans, after single or multiple doses, the pharmacokinetics of ketorolac is linear. Steady-state plasma levels are achieved after one day when administered 4 times daily. No changes were observed with long-term dosing. After a single intravenous dose, the volume of distribution is 0.25 l/kg, the half-life is 5 hours, and the clearance is 0.55 ml/min/kg. The main route of excretion of ketorolac and its metabolites (conjugates and p-hydroxymetabolites) is urine (91.4%), and the rest is excreted in feces. A diet rich in fat reduces the rate of absorption, but not the volume, while antacids do not affect the absorption of ketorolac.
Indications for use
Ketorol Insta, orally dispersible tablets, 10 mg, is used for the short-term treatment of acute pain (including postoperative pain) of moderate to severe intensity, only as a continuation of previous parenteral (intramuscular or intravenous) therapy in a hospital setting, if necessary. The total duration of parenteral and oral therapy with ketorolac should not exceed 5 days due to the possibility of increasing the frequency and severity of adverse reactions. Before taking Ketorol Ineta, consider the potential benefits and risks and options for using another drug. Use the lowest effective dose for the shortest possible time. Patients should be switched to alternative treatment as quickly as possible.
Directions for use and doses
The duration of the course of use of Ketorol Ineta should not exceed 5 days; long-term use, as well as oral administration at a dose of more than 40 mg per day, is not recommended. To reduce the risk of side effects, it is recommended to use the minimum effective dose for the minimum time required to relieve pain. From 17 to 64 years: 10 mg every 4-6 hours as needed. It is not recommended to use the drug in doses exceeding 40 mg per day. For patients receiving ketorolac parenterally and prescribed oral ketorolac tablets, the total combined daily dose should not exceed 90 mg (60 mg for the elderly, patients with impaired renal function, and patients weighing less than 50 mg), and The dosage of the oral form of the drug should not exceed 40 mg per day. Patients should be switched to oral administration of the drug as early as possible. Elderly patients Elderly patients have a higher risk of developing severe complications, particularly from the digestive tract. During treatment with NSAIDs, the patient's condition should be regularly monitored; a longer interval between doses of the drug is usually recommended, for example, 6-8 hours. Dispersible tablets can be used in patients with swallowing dysfunction. Can be taken without drinking water. The tablet is placed on the tongue, kept in the mouth until it is completely dissolved (do not chew), then swallowed.
Use during pregnancy and lactation
The safety of ketorolac during pregnancy has not been established. Given the known effect of NSAIDs on the cardiovascular system of the fetus (risk of premature closure of the ductus arteriosus), ketorolac is contraindicated during pregnancy, labor and childbirth. The onset of labor may be delayed and the duration prolonged, with an increased tendency for bleeding to occur in both mother and baby. Ketorolac is excreted into breast milk in small quantities, therefore Ketorol Insta is contraindicated during breastfeeding.
Precautionary measures
The maximum duration of use, including the use of parenteral forms, should not exceed 5 days. Gastrointestinal bleeding, ulceration and perforation. Gastrointestinal bleeding, ulceration or perforation, which may be fatal, has been reported with the use of NSAIDs at any time during treatment, with or without warning symptoms or in the case of a history of severe gastrointestinal disorders. The risk of developing severe gastrointestinal bleeding depends on the dosage of the drug. This, in particular, applies to elderly patients who use ketorolac in an average daily dose above 60 mg. For these patients, as well as for patients who are concomitantly using low doses of acetylsalicylic acid or other drugs that may increase gastrointestinal risk, combination treatment with protective agents (eg, misoprostol or proton pump inhibitors) should be considered. Ketorol Ineta should be used with caution in patients receiving concurrent medications that may increase the risk of ulceration or bleeding, such as oral corticosteroids, selective serotonin reuptake inhibitors, or antiplatelet agents such as acetylsalicylic acid. If gastrointestinal bleeding or ulceration occurs in patients receiving Ketorol Ineta, the course of treatment should be discontinued. Respiratory dysfunction. Caution is required when using the drug in patients with bronchial asthma (or with a history of asthma), since NSAIDs have been reported to accelerate the onset of bronchospasm in such patients. Effect on the kidneys. Prostaglandin biosynthesis inhibitors (including NSAIDs) have been reported to have nephrotoxic effects. The drug should be prescribed with caution to patients with impaired renal, cardiac, or liver function, since the use of NSAIDs may lead to deterioration of renal function. Patients with mildly impaired renal function are prescribed lower doses of ketorolac (those that do not exceed 60 mg per day intramuscularly or intravenously), and the renal condition of such patients should be carefully monitored. As with other drugs that inhibit prostaglandin synthesis, increases in serum urea, creatinine, and potassium have been reported during use of ketorolac tromethamine, which may occur after a single dose. Disorders of the cardiovascular system, kidneys and liver. The drug should be used with caution in patients with conditions that lead to a decrease in blood volume and/or renal blood flow, when renal prostaglandins play a supporting role in ensuring renal perfusion. In these patients, renal function should be monitored. The volume reduction should be corrected and serum urea and creatinine levels and the volume of urine excreted carefully monitored until the patient becomes normovolemic. In patients on renal dialysis, the clearance of ketorolac was reduced by approximately half the normal rate, and the terminal half-life was approximately tripled. Patients with impaired liver function due to cirrhosis did not have any clinically significant changes in ketorolac clearance or terminal half-life. Borderline increases in one or more liver function tests may occur. These abnormalities may be temporary, may remain unchanged, or may progress with continued treatment. If clinical signs and symptoms indicate the development of liver disease or if systemic manifestations are observed, Ketorol Ineta should be discontinued. Fluid retention and edema Fluid retention and edema have been reported during the use of ketorolac and should be used with caution in patients with cardiac decompensation, hypertension or similar conditions. Cardiovascular and cerebrovascular effects. There is currently insufficient information to assess such a risk for ketorolac tromethamine. Patients with uncontrolled hypertension, congestive heart failure, diagnosed coronary artery disease, peripheral arterial disease and/or cerebrovascular disease should be under medical supervision. Systemic lupus erythematosus and mixed connective tissue diseases. In patients with systemic lupus erythematosus and various mixed connective tissue diseases, the risk of developing aseptic meningitis increases. Dermatological. Ketorol Ineta should be discontinued at the first signs of a skin rash, damage to the mucous membranes or any other signs of hypersensitivity. Anaphylactic (anaphylactoid) reactions. As with the use of other NSAIDs, anaphylactic (anaphylactoid) reactions (including anaphylaxis, bronchospasm, hyperemia, rash, hypotension, laryngeal edema, Quincke's edema) may occur in patients who have no or no history of hypersensitivity to aspirin, other NSAIDs or ketorolac. This set of symptoms may also occur in individuals with a history of bronchospastic reactivity (eg, asthma) and nasal polyps. Anaphylactoid reactions, such as anaphylaxis, can be fatal. Therefore, ketorolac should not be taken by patients with a history of asthma and patients with complete or partial nasal polyp syndrome, angioedema and bronchospasm. If anaphylactoid reactions occur, seek emergency medical attention. Hematological effects. Patients with bleeding disorders should not be prescribed Ketorol Ineta. Patients receiving anticoagulant therapy may have an increased risk of bleeding if ketorolac is used concomitantly. Patients receiving other drugs that may affect the rate of bleeding control should be carefully monitored when ketorolac is prescribed to them. In controlled clinical trials, the incidence of significant postoperative bleeding was less than 1%. Ketorolac inhibits platelet aggregation and prolongs bleeding time. In patients with normal bleeding times, the duration of bleeding increased, but did not exceed the normal range of 2-11 minutes. In contrast to the long-term effects resulting from the use of acetylsalicylic acid, platelet function returns to normal within 24-48 hours after discontinuation of ketorolac. Ketorolac should not be prescribed to patients who have undergone surgery with a high risk of bleeding or incomplete control of bleeding. Caution should be used if mandatory bleeding control is critical. Ketorol Ineta is not an anesthetic and does not have sedative or anxiolytic properties; therefore, it is not recommended as a pre-operative sedative to maintain anesthesia.
Interaction with other drugs
Due to the possibility of side effects, ketorolac should not be prescribed with other NSAIDs, including selective cyclooxygenase-2 inhibitors, or in patients receiving acetylsalicylic acid, warfarin, lithium, probenecid, cyclosporine. NSAIDs should not be prescribed within 8-12 days after using mifepristone, since NSAIDs may weaken the effect of mifepristone. Medicines in combination with ketorolac should be prescribed with caution. In healthy normovolemic individuals, ketorolac reduces the diuretic effect of furosemide by approximately 20%; therefore, the drug is prescribed with particular caution to patients with cardiac decompensation. NSAIDs may worsen heart failure, reduce glomerular filtration rate, and increase plasma levels of cardiac glycosides when coadministered with cardiac glycosides. Ketorolac and other non-steroidal anti-inflammatory drugs may weaken the effect of antihypertensive drugs. In the case of simultaneous use of ketorolac with ACE inhibitors, there is an increased risk of renal dysfunction, especially in patients with a reduced blood volume in the body. There is a possible risk of nephrotoxicity if NSAIDs are prescribed with tacrolimus. Co-administration with diuretics may lead to a weakening of the diuretic effect and an increased risk of NSAID nephrotoxicity. As with all NSAIDs, caution is required when coadministering corticosteroids due to the increased risk of gastrointestinal ulceration or bleeding. There is an increased risk of gastrointestinal bleeding if NSAIDs are prescribed in combination with antiplatelet agents and selective serotonin reuptake inhibitors. Caution is advised if methotrexate is coadministered as some prostaglandin synthesis inhibitors have been reported to reduce the clearance of methotrexate and therefore possibly increase its toxicity. Patients taking NSAIDs and quinolones may have an increased risk of developing seizures. Concomitant use of NSAIDs with zidovudine leads to an increased risk of hematological toxicity. There is an increased risk of hemarthrosis and hematoma in HIV-infected people who have hemophilia and who are treated with both zidovudine and ibuprofen. The following drugs are unlikely to interact with ketorolac. Ketorolac did not affect the binding of digoxin to plasma protein. In vitro studies indicate that at therapeutic salicylate concentrations (300 mcg/mL) and higher, ketorolac binding was reduced by approximately 99.2% to 97.5%. Therapeutic concentrations of digoxin, warfarin, paracetamol, phenytoin and tolbutamide did not affect the binding of ketorolac to plasma protein. Because ketorolac is a highly active drug and its available plasma concentration is low, it is not expected to significantly replace other drugs that bind to plasma proteins. There is no evidence from animal or human studies that ketorolac tromethamine induces or inhibits liver enzymes that are capable of metabolizing it or other drugs. Therefore, ketorolac is not expected to be metabolized by it or other drugs. Therefore, ketorolac is not expected to alter the pharmacokinetics of other drugs.
Contraindications
Hypersensitivity to ketorolac or any component of the drug; patients with an active peptic ulcer, with recent gastrointestinal bleeding or perforation, with a history of peptic ulcer disease or gastrointestinal bleeding; bronchial asthma, rhinitis, angioedema or urticaria caused by the use of acetylsalicylic acid or other non-steroidal anti-inflammatory drugs (due to the possibility of severe anaphylactic reactions); do not use as an analgesic before and during surgery; complete or partial nasal polyp syndrome, angioedema or bronchospasm; do not use in patients who have had surgery with a high risk of hemorrhage or incomplete bleeding control and in patients who receive anticoagulants, including low doses of heparin (2500-5000 units every 12 hours); hepatic or moderate to severe renal failure (serum creatinine concentration more than 160 µmol/l; suspected or confirmed cerebrovascular bleeding, bleeding diathesis, including coagulation disorders and a high risk of bleeding; concomitant treatment with other non-steroidal anti-inflammatory drugs (NSAIDs) (including selective inhibitors cyclooxygenase), acetylsalicylic acid, warfarin, pentoxifylline, probenecid or lithium salts; hypovolemia, dehydration; pregnancy, labor, childbirth and breastfeeding; children under 16 years of age; erosive and ulcerative lesions and inflammatory diseases of the oral cavity.
Compound
Active ingredient: ketorolac tromethamine – 10 mg; excipients - microcrystalline cellulose, hydrated colloidal silicon dioxide; antioxidant butylated hydroxyanisole, mannitol crospovidone, sucralose, mint flavor, quinoline yellow dye (E104), magnesium stearate.
Overdose
Symptoms: headache, nausea, vomiting, epigastric pain, gastrointestinal bleeding; rarely - diarrhea, disorientation, agitation, coma, drowsiness, dizziness, ringing in the ears, loss of consciousness, sometimes convulsions. In cases of severe poisoning, acute renal failure and liver damage are possible. Anaphylactoid reactions have been observed after therapeutic use of NSAIDs, which may occur after overdose. Treatment: gastric lavage, use of activated carbon. It is necessary to ensure sufficient diuresis. Kidney and liver function should be carefully monitored. Patients should be monitored for at least 4 hours after ingestion of a potentially toxic amount. Frequent or prolonged seizures should be treated with intravenous diazepam. Other measures may be prescribed depending on the patient's clinical condition. Therapy is symptomatic.
Side effect
From the digestive tract: peptic ulcer, perforation or gastrointestinal bleeding, sometimes fatal (especially in the elderly), nausea, dyspepsia, gastrointestinal pain, abdominal discomfort, hematemesis, gastritis, esophagitis, diarrhea, belching, constipation, flatulence, feeling of fullness in the stomach, melena, rectal bleeding, ulcerative stomatitis, vomiting, hemorrhage, perforation, pancreatitis, exacerbation of colitis and Crohn's disease. From the central nervous system: anxiety, blurred vision, optic neuritis, drowsiness, dizziness, increased sweating, dry mouth, nervousness, paresthesia, functional disorders, depression, euphoria, convulsions, inability to concentrate, insomnia, increased fatigue, agitation, vertigo, impaired taste and vision, myalgia, unusual dreams, confusion, hallucinations, hyperkinesia, hearing loss, tinnitus, psychotic reactions, thinking disorders. Infectious diseases: aseptic meningitis (especially in patients with autoimmune diseases such as systemic lupus erythematosus, mixed connective tissue disease), stiff neck, headache, nausea, vomiting, fever, confusion. Metabolism and nutrition: anorexia, hyponatremia, hyperkalemia. From the urinary system, increased urinary frequency, oliguria, acute renal failure, hemolytic uremic syndrome, flank pain (with/without hematuria), increased serum urea and creatinine, interstitial nephritis, urinary retention, nephrotic syndrome, infertility, renal failure. From the liver: impaired liver function, hepatitis, jaundice and liver failure, increased functional tests. From the cardiovascular system: flushing of the face, bradycardia, pallor, hypertension, hypotension, palpitation, chest pain, edema, heart failure. Data from clinical and epidemiological studies indicate that the use of certain NSAIDs, especially in high doses and for long periods of time, may be associated with an increased risk of arterial thromboembolic complications (myocardial infarction or stroke). From the respiratory system: nosebleeds, shortness of breath, asthma, pulmonary edema. From the blood system: purpura, thrombocytopenia, neutropenia, agranulocytosis, aplastic and hemolytic anemia. Skin: itching, urticaria, photosensitivity of the skin, Lyell's syndrome, bullous reactions, including Stevens-Johnson syndrome and toxic epidermal necrolysis (very rare), exfoliative dermatitis, maculopapular rashes. Effect on reproductive function: the use of ketorolac, like any drug that inhibits the synthesis of cyclooxygenase/prostaglandin, can cause infertility. Hypersensitivity: Hypersensitivity reactions have been reported, which include nonspecific allergic reactions and anaphylaxis, respiratory tract reactivity including asthma, worsening asthma, bronchospasm, laryngeal edema or shortness of breath, and a variety of skin disorders that include various types of rashes, itching , urticaria, purpura, angioedema, and in isolated cases, exfoliative and bullous dermatitis (including epidermal necrolysis and erythema multiforme). Such reactions may occur in patients without or with known hypersensitivity to ketorolac or other non-steroidal anti-inflammatory drugs. They may also occur in individuals with a history of angioedema, bronchospastic reactivity (eg, asthma, and nasal polyps). Anaphylactoid reactions, such as anaphylaxis, can be fatal. Others: postoperative bleeding from a wound, hematoma, prolongation of bleeding, asthenia, edema, weight gain, increased body temperature, excessive thirst, fatigue, malaise, fever, chest pain. To prevent possible side effects, one should strive to use the minimum effective doses of the drug, strictly adhere to the established dosages and administration regimens, take into account the patient’s condition (age, renal function, state of the gastrointestinal tract, water-electrolyte metabolism and hemostatic system), as well as possible drug interactions with combination therapy.
Storage conditions
Store at a temperature not exceeding 25°C. Keep out of the reach of children.
Types of strong painkillers for tooth pain
For toothache, different types of painkillers can be used. Let's list:
- Tablets are used to influence the pain epicenter. They can dull the discomfort for several hours.
- The powder is gentle on the gastrointestinal tract and has anti-inflammatory and antipyretic effects.
- Toothpaste helps with increased sensitivity of tooth enamel. That is, if there is caries, pain relief will not occur. In the first case, it is enough to brush your teeth regularly.
- The gel is also able to temporarily relieve toothache due to its local anesthetic effect.
About contraindications
Any doctor will tell you that any painkillers for toothache also have a negative effect on the body. This is due to the fact that they involve the functioning of the gastrointestinal tract, kidneys, liver, cardiac and nervous systems. But side effects are observed when pills and other types of drugs are taken incorrectly or when treatment regimens are not followed.
Main contraindications for toothache tablets:
- individual intolerance;
- pregnancy;
- lactation period;
- ulcer;
- heart failure;
- kidney or liver problems;
- asthma;
- problems with the circulatory system;
- childhood.
In fact, the body can react individually to the action of substances, so before taking any painkillers you should read the instructions, and, if possible, also consult a specialist.